Hazardous Products Regulations (SOR/2015-17)
Full Document:
- HTMLFull Document: Hazardous Products Regulations (Accessibility Buttons available) |
- XMLFull Document: Hazardous Products Regulations [569 KB] |
- PDFFull Document: Hazardous Products Regulations [1196 KB]
Regulations are current to 2026-05-26 and last amended on 2022-12-15. Previous Versions
PART 8Health Hazard Classes
SUBPART 1Acute Toxicity
Definitions
Marginal note:Definitions
8.1 The following definitions apply in this Subpart.
- acute toxicant
acute toxicant means a mixture or substance that is liable to cause acute toxicity, or a mixture or substance that, upon contact with water, releases a gaseous substance that is liable to cause acute toxicity. (toxique aigu)
- acute toxicity
acute toxicity refers to serious adverse health effects, including death, occurring following
(a) oral or dermal administration of a single dose of a mixture or substance, or multiple doses given within 24 hours; or
(b) an inhalation exposure to a mixture or substance of four hours or of a duration that is converted to four hours in accordance with subsection 8.1.1(4). (toxicité aiguë)
- dust
dust means solid particles that are suspended in a gas, usually air. (poussières)
- mist
mist means liquid droplets that are suspended in the air. (brouillard)
Classification in a Category of the Class
Classification of Substances
Marginal note:LD50 or LC50 — associated range
8.1.1 (1) An acute toxicant that is a substance is classified, with respect to each applicable route of exposure, in a category of this hazard class in accordance with the tables to subsection (3) if it has an LD50 by the oral or dermal exposure route, or an LC50 by the inhalation exposure route, that falls into one of the ranges indicated in the applicable table to that subsection.
Marginal note:Contact with water — gaseous substance
(2) If an acute toxicant that is a substance is not classified in accordance with subsection (1) with respect to the inhalation route of exposure in a category of this hazard class in accordance with Table 3 to subsection (3) and, upon contact with water, the acute toxicant releases a gaseous substance that has an LC50 that falls into one of the ranges indicated in that table, it is classified in accordance with that table with regard to that range.
Marginal note:LD50 or LC50 not available
(3) If an LD50 by the oral or dermal exposure route or an LC50 by the inhalation exposure route is not available, an acute toxicity point estimate must be established in accordance with the table to section 8.1.7, and the acute toxicant must be classified based on that acute toxicity point estimate, with respect to each applicable route of exposure, in a category of this hazard class in accordance with the following tables:
TABLE 1
Oral Exposure Route
Column 1 Column 2 Item Category Ranges for LD50 or for Acute Toxicity Point Estimates (mg/kg body weight) 1 Acute Toxicity (Oral) — Category 1 ≤ 5 2 Acute Toxicity (Oral) — Category 2 > 5 and ≤ 50 3 Acute Toxicity (Oral) — Category 3 > 50 and ≤ 300 4 Acute Toxicity (Oral) — Category 4 > 300 and ≤ 2000 TABLE 2
Dermal Exposure Route
Column 1 Column 2 Item Category Ranges for LD50 or for Acute Toxicity Point Estimates (mg/kg body weight ) 1 Acute Toxicity (Dermal) — Category 1 ≤ 50 2 Acute Toxicity (Dermal) — Category 2 > 50 and ≤ 200 3 Acute Toxicity (Dermal) — Category 3 > 200 and ≤ 1000 4 Acute Toxicity (Dermal) — Category 4 > 1000 and ≤ 2000 TABLE 3
Inhalation Exposure Route
Column 1 Column 2 Column 3 Column 4 Ranges for LC50 or for Acute Toxicity Point Estimates Item Category Gases (ppmV) Vapours (mg/l) Dusts and Mists (mg/l) 1 Acute Toxicity (Inhalation) — Category 1 ≤ 100 ≤ 0.5 ≤ 0.05 2 Acute Toxicity (Inhalation) — Category 2 > 100 and ≤ 500 > 0.5 and ≤ 2 > 0.05 and ≤ 0.5 3 Acute Toxicity (Inhalation) — Category 3 > 500 and ≤ 2500 > 2 and ≤ 10 > 0.5 and ≤ 1 4 Acute Toxicity (Inhalation) — Category 4 > 2500 and ≤ 20 000 > 10 and ≤ 20 > 1 and ≤ 5 Marginal note:One-hour exposure period
(4) For the purposes of Table 3 to subsection (3), the LC50 is based on a four-hour exposure period. If existing acute inhalation toxicity data have been generated according to a one-hour exposure period, the LC50 for gases and vapours must be divided by two, and the LC50 for dusts and mists must be divided by four.
Classification of Mixtures
Marginal note:Order of provisions
8.1.2 (1) The classification of a mixture as an acute toxicant in a category of this hazard class must proceed in accordance with the order of sections 8.1.3 to 8.1.6.
Marginal note:Concentrations for the purpose of classification
(2) Only ingredients present at concentrations equal to or greater than the concentration limit of 1.0% — w/w for solids, liquids, dusts, mists and vapours and v/v for gases — must be considered for the purpose of classification.
Marginal note:Data available for mixture as a whole
8.1.3 If data of the types referred to in subparagraphs 2.1(a)(i) to (iv) are available for the mixture as a whole, the mixture must be classified as an acute toxicant in accordance with section 8.1.1.
Marginal note:Data available for use of bridging principles
8.1.4 If data are available to enable the characterization of the mixture as an acute toxicant, in accordance with the bridging principles referred to in subsections 2.3(3) to (8), the mixture must be classified in a category of this hazard class in accordance with those subsections.
Marginal note:Data available for all ingredients
8.1.5 If data are available for all ingredients in the mixture, the mixture must be classified as an acute toxicant in accordance with section 8.1.1 using the ATE of the mixture that is determined in respect of each applicable route of exposure by the following formula:
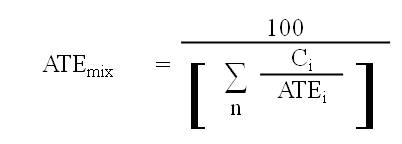
where
- ATEmix
- is the ATE of the mixture determined using this formula;
- Ci
- is the concentration of ingredient i;
- n
- is the number of ingredients and i is running from 1 to n;
- ATEi
- is the ATE of ingredient i, which is either
(a) the LD50 or the LC50 based on or converted to a four-hour exposure period, for i, or
(b) if the LD50 or the LC50 is unavailable, the acute toxicity point estimate established for i in accordance with the table to section 8.1.7; and
- i
- is each ingredient in the mixture with
(a) an ATE within the ranges set out in the applicable table to subsection 8.1.1(3),
(b) an oral or dermal LD50 greater than 2000 mg/kg body weight but less than or equal to 5000 mg/kg body weight, or
(c) an LC50 based on or converted to a four-hour exposure period within a range having an amplitude comparable to the one in paragraph (b).
Marginal note:Data not available for all ingredients
8.1.6 If the ATE is not available for one or more ingredients of the mixture, the mixture must be classified as an acute toxicant in accordance with section 8.1.1 using the ATE of the mixture that is determined in respect of each applicable route of exposure according to the following:
(a) if data permit the ATE to be estimated for each of those ingredients in accordance with established scientific principles, the formula in section 8.1.5 must be used;
(b) if data do not permit the ATE to be estimated for an ingredient in accordance with established scientific principles, and the concentration of the ingredient in the mixture is equal to or greater than the concentration limit of 1.0%, the mixture is classified based only on the ingredients having an ATE, such that
(i) if the total concentration of all ingredients with unknown acute toxicity is less than or equal to 10.0% of the mixture, the formula in section 8.1.5 must be used, or
(ii) if the total concentration of all ingredients with unknown acute toxicity is greater than 10.0% of the mixture, the following formula must be used:
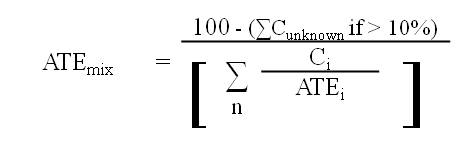
where
- ATEmix
- is the ATE of the mixture determined using this formula,
- Ci
- is the concentration of ingredient i,
- Cunknown
- is the concentration of ingredients i with unknown ATE values,
- n
- is the number of ingredients and i is running from 1 to n,
- ATEi
- is the ATE of ingredient i, which is either
(a) the LD50 or the LC50 based on or converted to a four-hour exposure period, for i, or
(b) if the LD50 or the LC50 is unavailable, the acute toxicity point estimate established for i in accordance with the table to section 8.1.7, and
- i
- is each ingredient in the mixture with
(a) an ATE within the ranges set out in the applicable table to subsection 8.1.1(3),
(b) an oral or dermal LD50 greater than 2000 mg/kg body weight but less than or equal to 5000 mg/kg body weight, or
(c) an LC50 based on or converted to a four-hour exposure period within a range having an amplitude comparable to the one in paragraph (b).
Marginal note:Conversion from range to point estimate
8.1.7 (1) If a formula in section 8.1.5 or 8.1.6 is used, an acute toxicity point estimate must be determined, in accordance with the table to subsection (2), for each ingredient for which only that ingredient’s classification category or experimentally obtained acute toxicity range is available.
Marginal note:More than one range
(2) If the experimentally obtained acute toxicity range for an ingredient does not fall entirely within any of the ranges set out in column 2 of the following table, the converted acute toxicity point estimate for that ingredient for the purposes of column 3 is the lowest value of the experimentally obtained acute toxicity range.
TABLE
Column 1 Column 2 Column 3 Item Exposure Routes Classification Category and Associated Experimentally Obtained Acute Toxicity Range Minimum and Maximum Values Converted Acute Toxicity Point Estimate 1 Oral (mg/kg body weight) - 0 < Category 1 ≤ 5
- 5 < Category 2 ≤ 50
- 50 < Category 3 ≤ 300
- 300 < Category 4 ≤ 2000
- 0.5
- 5
- 100
- 500
2 Dermal (mg/kg body weight) - 0 < Category 1 ≤ 50
- 50 < Category 2 ≤ 200
- 200 < Category 3 ≤ 1000
- 1000 < Category 4 ≤ 2000
- 5
- 50
- 300
- 1100
3 Inhalation (gases) (ppmV) - 0 < Category 1 ≤ 100
- 100 < Category 2 ≤ 500
- 500 < Category 3 ≤ 2500
- 2500 < Category 4 ≤ 20 000
- 10
- 100
- 700
- 4500
4 Inhalation (vapours) (mg/l) - 0 < Category 1 ≤ 0.5
- 0.5 < Category 2 ≤ 2.0
- 2.0 < Category 3 ≤ 10.0
- 10.0 < Category 4 ≤ 20.0
- 0.05
- 0.5
- 3
- 11
5 Inhalation (dust/mist) (mg/l) - 0 < Category 1 ≤ 0.05
- 0.05 < Category 2 ≤ 0.5
- 0.5 < Category 3 ≤ 1.0
- 1.0 < Category 4 ≤ 5.0
- 0.005
- 0.05
- 0.5
- 1.5
Page Details
- Date modified: